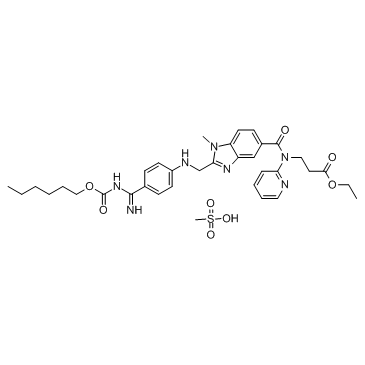
ʻO Dabigatran Etexilate Mesylate
wehewehe
ʻO Dabigatran etexilate mesylate (BIBR 1048MS) kahi mea hoʻohana waha o Dabigatran. He hopena anticoagulant ʻo Dabigatran etexilate mesylate a hoʻohana ʻia no ka prophylaxis o ka venousthromboembolism a me ka hahau ma muli o ka fibrillation atrial.
Kāpae
Hōʻike: IC50 Waiwai: 4.5nM (Ki); 10nM(Thrombin-induced platelet aggregation) [1] ʻO Dabigatran he mea hiki ke hoʻohuli a koho, pololei thrombin inhibitor (DTI) e hele nei i ka hoʻomohala ʻana i nā lāʻau lapaʻau e like me kāna prodrug hoʻoikaika waha, dabigatran etexilate. in vitro: Dabigatran selectively and reversibly inhibited human thrombin (Ki: 4.5 nM) a me ka thrombin-induced platelet aggregation (IC(50): 10 nM), me ka hōʻike ʻole i ka hopena inhibitory ma nā mea hoʻoulu platelet-stimulating agents.Thrombin generation in platelet -ʻo ka plasma maikaʻi ʻole (PPP), i ana ʻia e like me ka hiki ʻana o ka thrombin endogenous (ETP) i kaohi ʻia ma muli o ka hilinaʻi. (IC(50): 0.56 microM). Ua hōʻike ʻo Dabigatran i nā hopena anticoagulant pili i ka ʻike i nā ʻano like ʻole i loko o ka vitro, e pālua i ka manawa o ka thromboplastin partial (aPTT), ka manawa prothrombin (PT) a me ka manawa clotting ecarin (ECT) i loko o ka PPP kanaka ma nā ʻano o 0.23, 0.83 a me 0.18 microM. 1]. in vivo: Ua hoʻolōʻihi ʻia ʻo Dabigatran i ka nui o ka aPTT ma hope o ka lawelawe intravenous i nā ʻiole (0.3, 1 a me 3 mg / kg) a me nā monkey rhesus (0.15, 0.3 a me 0.6 mg / kg). Ua ʻike ʻia nā hopena anticoagulant e pili ana i ka dosis a me ka manawa me ka dabigatran etexilate i hāʻawi waha ʻia i nā ʻiole ʻike (10, 20 a me 50 mg / kg) a i ʻole rhesus monkeys (1, 2.5 a i ʻole 5 mg / kg), me nā hopena kiʻekiʻe loa i ʻike ʻia ma waena o 30 a me 120. min ma hope o ka lawelawe ʻana, i kēlā me kēia [1]. ʻO nā poʻe maʻi i mālama ʻia me ka dabigatran etexilate ua liʻiliʻi ka liʻiliʻi o nā hahau ischemic (3.74 dabigatran etexilate vs 3.97 warfarin) a me ka liʻiliʻi o nā hemorrhages intracranial hui pū ʻia a me nā hahau hemorrhagic (0.43 dabigatran etexilate vs 0.99 warfarin) i kēlā me kēia 120 mau makahiki maʻi. Ka ho'āʻo hoʻokolohua: He loiloi o nā Pharmacokinetics a me Pharmacodynamics o Oral Dabigatran Etexilate i nā maʻi Hemodialysis. Māhele1
Waihona
| Pauda | -20°C | 3 makahiki |
| 4°C | 2 makahiki | |
| I ka solvent | -80°C | 6 mahina |
| -20°C | 1 mahina |
Hoao Hoao
| Helu NCT | Kākoʻo | Kūlana | lā hoʻomaka | Māhele |
| NCT02170792 | ʻO Boehringer Ingelheim | Ola ola | Pepeluali 2001 | Māhele 1 |
| NCT02170974 | ʻO Boehringer Ingelheim | Ola ola | Iulai 2004 | Māhele 1 |
| NCT02170831 | ʻO Boehringer Ingelheim | Ola ola | Mei 1999 | Māhele 1 |
| NCT02170805 | ʻO Boehringer Ingelheim | Ola ola | ʻApelila 2001 | Māhele 1 |
| NCT02170610 | ʻO Boehringer Ingelheim | Ola ola | Malaki 2002 | Māhele 1 |
| NCT02170909 | ʻO Boehringer Ingelheim | Ola ola | Kekemapa 2004 | Māhele 1 |
| NCT02171000 | ʻO Boehringer Ingelheim | Ola ola | ʻApelila 2005 | Māhele 1 |
| NCT02170844 | ʻO Boehringer Ingelheim | Ola ola | Iune 2004 | Māhele 1 |
| NCT02170584 | ʻO Boehringer Ingelheim | Ola ola | Ianuali 2001 | Māhele 1 |
| NCT02170935 | ʻO Boehringer Ingelheim | ʻO ka Thromboembolism Venous | ʻApelila 2002 | Māhele 2 |
| NCT02170636 | ʻO Boehringer Ingelheim | Ola ola | Ianuali 2002 | Māhele 1 |
| NCT02170766 | ʻO Boehringer Ingelheim | Ola ola | ʻOkakopa 2000 | Māhele 1 |
| NCT02171442 | ʻO Boehringer Ingelheim | Ola ola | ʻApelila 2002 | Māhele 1 |
| NCT02170896 | ʻO Boehringer Ingelheim | Ola ola | ʻOkakopa 2001 | Māhele 1 |
| NCT02173730 | ʻO Boehringer Ingelheim | Ola ola | Nowemapa 2002 | Māhele 1 |
| NCT02170623 | ʻO Boehringer Ingelheim | Ola ola | Pepeluali 2002 | Māhele 1 |
| NCT02170116 | ʻO Boehringer Ingelheim | Ola ola | Nowemapa 1998 | Māhele 1 |
| NCT02170597 | ʻO Boehringer Ingelheim | Ola ola | ʻAukake 2003 | Māhele 1 |
| NCT01225822 | ʻO Boehringer Ingelheim | ʻO ka Thromboembolism Venous | Nowemapa 2002 | Māhele 2 |
| NCT02170701 | ʻO Boehringer Ingelheim | ʻO ka Thromboembolism Venous | ʻOkakopa 2000 | Māhele 2 |
| NCT02170740 | ʻO Boehringer Ingelheim | Ola ola | Nowemapa 1999 | Māhele 1 |
| NCT02170922 | ʻO Boehringer Ingelheim | Ola ola | Iulai 1999 | Māhele 1 |
ʻO ke kinona





Manaʻo18Nā papahana Loiloi Kūlike Kūlike i ʻae ʻia4, a6apono ʻia nā papahana.

Ua hoʻokumu ka ʻōnaehana hoʻokele maikaʻi honua kiʻekiʻe i ke kumu kūʻai.

Holo ka nānā ʻana i ka maikaʻi ma ke ola holoʻokoʻa o ka huahana e hōʻoia i ka maikaʻi a me ka hopena therapeutic.

Kākoʻo ka hui ʻoihana Regulatory Affairs i nā koi maikaʻi i ka wā o ke noi a me ke kākau inoa ʻana.


Kolea Countec Bottled Packaging Line


ʻO Taiwan CVC Bottled Packaging Line


Italia CAM Board Packaging Line

Mekini Fette Compacting German

ʻO Iapana Viswill Tablet ʻIke

Keena Mana o DCS







