Nūhou
-

Pono ka maʻi puʻuwai i kahi lāʻau hou - Vericiguat
ʻO ka hāʻule ʻana o ka naʻau me ka haʻihaʻi ejection hoʻemi ʻia (HFrEF) he ʻano nui o ka puʻuwai puʻuwai, a ua hōʻike ka China HF Study he 42% o ka pau ʻana o ka naʻau ma Kina he HFrEF, ʻoiai ua loaʻa kekahi mau papa lapaʻau maʻamau no HFrEF a ua hōʻemi i ka pilikia. o...Heluhelu hou aku -

ʻO ka lāʻau lapaʻau no ka mālama ʻana i ka myelofibrosis: Ruxolitinib
Ua kapa ʻia ʻo Myelofibrosis (MF) he myelofibrosis. He mai laha loa ia. A ʻaʻole ʻike ʻia ke kumu o kona pathogenesis. ʻO nā hiʻohiʻona maʻamau o ke koko ʻulaʻula juvenile a me ka anemia granulocytic juvenile me ka helu kiʻekiʻe o ka waimaka ʻulaʻula ʻulaʻula ...Heluhelu hou aku -

Pono ʻoe e ʻike i kēia mau helu 3 e pili ana i rivaroxaban
Ma ke ʻano he anticoagulant waha hou, ua hoʻohana nui ʻia ʻo rivaroxaban i ka pale ʻana a me ka mālama ʻana i ka maʻi thromboembolic venous a me ka pale ʻana i ka hahau i ka fibrillation atrial non-valvular. I mea e hoʻohana pono ai iā rivaroxaban, pono ʻoe e ʻike i kēia mau helu 3.Heluhelu hou aku -

Ua loaʻa i ka Changzhou Pharmaceutical ka ʻae ʻia e hana i nā Capsules Lenalidomide
Ua loaʻa iā Changzhou Pharmaceutical Factory Ltd., kahi lālā o Shanghai Pharmaceutical Holdings, ka Palapala Hoʻopaʻa Hoʻopaʻa Lapaʻau (Certificate No. 2021S01077, 2021S01078, 2021S01079) i hoʻopuka ʻia e ka State Drug Administration no Lenalidomide Capsules (Specification 5mg, ...Heluhelu hou aku -

He aha nā mea e mālama ai no nā papa rivaroxaban?
ʻO Rivaroxaban, ma ke ʻano he anticoagulant waha hou, ua hoʻohana nui ʻia i ka pale ʻana a me ka mālama ʻana i nā maʻi thromboembolic venous. He aha kaʻu e pono ai e hoʻolohe i ka lawe ʻana iā rivaroxaban? ʻAʻole e like me warfarin, ʻaʻole koi ʻo rivaroxaban i ka nānā ʻana i ke koko koko i hōʻike ʻia ...Heluhelu hou aku -
2021 Nā ʻae ʻana i ka lāʻau lapaʻau hou a FDA 1Q-3Q
Hoʻoikaika ka hana hou i ka holomua. I ka wā e pili ana i ka hana hou i ka hoʻomohala ʻana i nā lāʻau lapaʻau hou a me nā huahana biological therapeutic, kākoʻo ka FDA's Center for Drug Evaluation and Research (CDER) i ka ʻoihana lāʻau lapaʻau i kēlā me kēia pae o ke kaʻina hana. Me kona hoʻomaopopo ʻana i ...Heluhelu hou aku -

Nā hana hou o Sugammadex Sodium i ka wā ala o ka anesthesia
ʻO Sugammadex Sodium kahi mea kūʻē kūʻē o nā mea hoʻomaha hoʻomaha hoʻomaha non-depolarizing (myorelaxants), i hōʻike mua ʻia i nā kānaka i ka makahiki 2005 a ua hoʻohana ʻia i ka lāʻau lapaʻau ma ʻEulopa, ʻAmelika a me Iapana. Hoʻohālikelike ʻia me nā lāʻau anticholinesterase kuʻuna ...Heluhelu hou aku -

ʻO wai nā maʻi maʻi thalidomide kūpono i ka mālama ʻana!
He mea maikaʻi ʻo Thalidomide i ka mālama ʻana i kēia mau ʻōpū! 1. Ma kahi e hiki ai ke hoʻohana ʻia ka thalidomide paʻa. 1.1. ka maʻi ʻaʻai māmā. 1.2. Ka maʻi maʻi prostate. 1.3. nodal rectal cancer. 1.4. hepatocellular carcinoma. 1.5. Ka maʻi maʻi ʻōpū. ...Heluhelu hou aku -

Tofacitinib Citrate
ʻO Tofacitinib citrate kahi lāʻau lapaʻau (inoa kālepa Xeljanz) i hoʻokumu mua ʻia e Pfizer no kahi papa o ka waha o Janus kinase (JAK) inhibitors. Hiki iā ia ke hoʻokaʻawale i ka JAK kinase, kaohi i nā ala JAK / STAT, a ma laila e kāohi ai i ka transduction hōʻailona cell a me ka hōʻike gen pili a me ka hoʻōla ...Heluhelu hou aku -
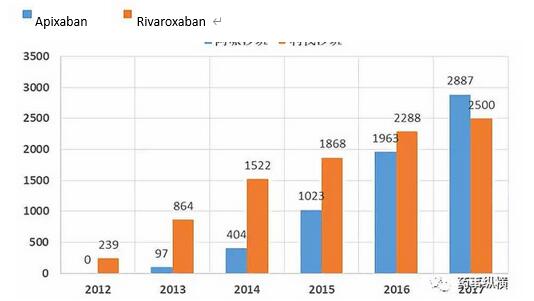
ʻO Apixaban a me Rivaroxaban
I nā makahiki i hala iho nei, ua ulu wikiwiki ka kūʻai ʻana o apixaban, a ua ʻoi aku ka mākeke honua ma mua o rivaroxaban. No ka mea ʻoi aku ka maikaʻi o Eliquis (apixaban) ma mua o warfarin i ka pale ʻana i ka hahau a me ke koko, a ʻo Xarelto (Rivaroxaban) wale nō i hōʻike i ka haʻahaʻa ʻole. Eia kekahi, ʻaʻole ʻo Apixaban ...Heluhelu hou aku -

Hōʻike ʻia ʻo Guangzhou API ma 2021
ʻO ka 86th China International Pharmaceutical Raw Materials/Intermediates/Packaging/Equipment Fair (API China no ka pōkole) Mea hoʻonohonoho: Reed Sinopharm Exhibition Co., Ltd. Ka manawa hōʻike: Mei 26-28, 2021 Wahi: China Import and Export Fair Complex (Guangzhou) Pākē hōʻike: 60,000 square meters Ex...Heluhelu hou aku -
Obeticholic acid
Ma Iune 29, ua hoʻolaha ʻo Intercept Pharmaceuticals ua loaʻa iā ia kahi noi lāʻau hou piha mai ka US FDA e pili ana i kāna FXR agonist obeticholic acid (OCA) no ka fibrosis i hoʻokumu ʻia e ka non-alcoholic steatohepatitis (NASH) pane pane (CRL). Ua ʻōlelo ka FDA ma ka CRL e pili ana i ka ʻikepili ...Heluhelu hou aku
